 Our research works will be introduced a little bit in detail.
Our research works will be introduced a little bit in detail.
Organic materials become magnets !
ü@It is usually believed that organic materials are insulators and diamagnets. However it is possible to bestow electrical conductivities and ferromagnetic properties to organic materials by appropriate molecular and crystallographic designs. We are making efforts to break the common senses, and to reveal the truth of nature.
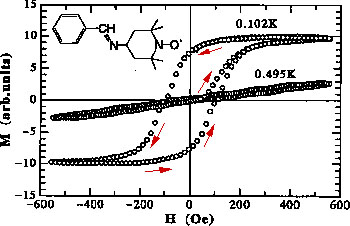
ü@The following is an example of an "organic magnet" composed of non-metallic elements such as carbon, hydrogen, nitrogen, and oxygen. It was the third example of the organic magnet developed in the world. The origin of magnetism, i. e., electron spin, comes from an organic radical. We utilized radicals sufficiently stable in the air and at room temperatures.
The following figure shows magnetization hysteresis curve of a genuine organic material, and this is characteristic of a ferromagnet.
What is a chiral magnetüH
ü@Molecular magnets possessing chiral crystal and magnetic structures are expected to show interesting solid-state properties such as magneto-chiral dichroism.
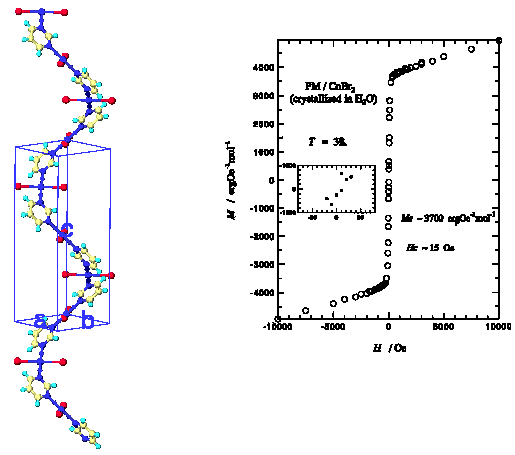
The following is a crystal structure and a magnetization curve of the chiral magnet, (pyrimidine)2CoBr2. Pyrimidine and CoBr2 constitute one-dimensional chiral and helical structures. The magnetization curve is typical of a weak-ferromagnet.
Is it possible to tune magnetic properties by means of supramolecular chemistryüH
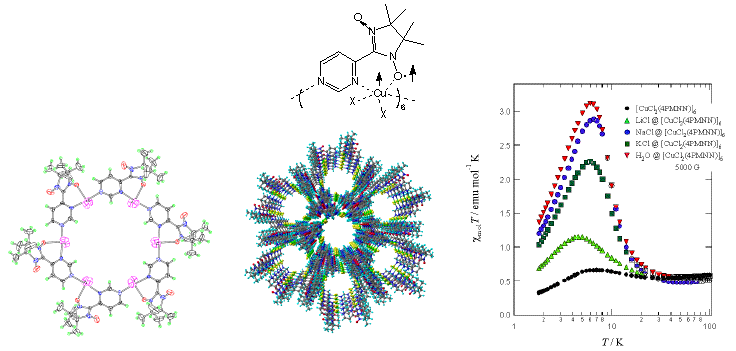
ü@We have synthesized 4NNPM which has a stable radical group (nitronyl nitroxide (NN)) at 4-position of pyrimidine. 4NNPM is regarded as a ligand possessing an electron spin. It gives a hexamer with CuX2 (X = Cl, Br) to form a hexagonal supramolecule, and it stacks to form a one-dimensional cavity with ca. 11
ü diameter. Ferromagnetic interaction is operative among Cu(II) and NN spins in the cavity direction. This ferromagnetic interaction was found to be greatly enhanced by encapsulation of alkali halides or water. The effect of water is noticeable; the magnetic property returns to the initial one after elimination of water. This system is a rare example of realizing the regulation of magnetic properties by encapsulation of molecules.
News Paper
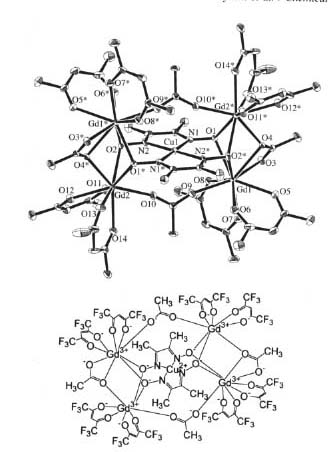
Super high-spin molecule with S = 27/2 !
ü@Single-molecule magnets (SMMs) have attracted much attention because it may be applied to memory/display/computing devices in nanotechnology. We have synthesized a Gd4Cu complex shown below. In this complex, four Gd spins are parallel each other (S = 4 x 7/2 = 14), and Gd spins and Cu spin (S = 1/2) are antiparallel. Therefore, the total spin of this molecule is 14 - 1/2 = 27/2.
What is a "single-molecule magnet" ?
SMMs show magnetic hysteresis from the single-molecular origin. Synthetic strategies of SMMs are:
1. Molecule with a large magnetic moment
2. Molecule with a large magnetic anisotropy
3. Molecule with very small intermolecular magnetic interactions
Dy4Cu and Tb4Cu complexes are isomorphous to Gd4Cu. They have almost no intermolecular magnetic interaction. In spite of this, its magnetization curves measured at 0.5 K show characteristic hysteresis (shown below), which means this complex to be a SMM.
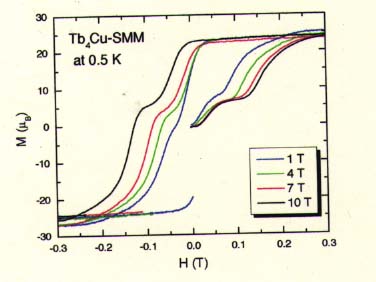
Coercivity World Record !
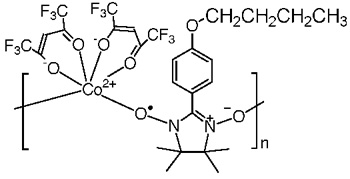
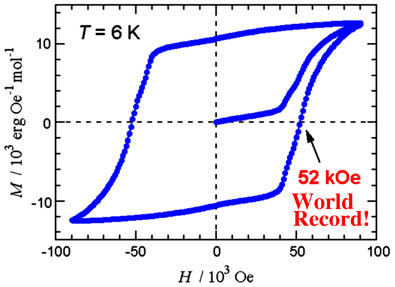
A radical-cobalt hybrid system CoBPNN was prepared. We found the coercive field of CoBPNN was 52 kOe (4.1 MA m-1) at 6 K as shown below, which is much larger than those of commercial permanent magnets SmCo5 (44 kOe at room temp.) and Nd2Fe14B (19 kOe at room temp.). To the best of our knowledge, this compound enjoys to be a record holder of the largest coercive field among hard magnets ever known.
 Back to Home Page
Back to Home Page
 Our research works will be introduced a little bit in detail.
Our research works will be introduced a little bit in detail. Our research works will be introduced a little bit in detail.
Our research works will be introduced a little bit in detail.





